GI Esophageal Cancer demo-test

The KEYNOTE-590 study

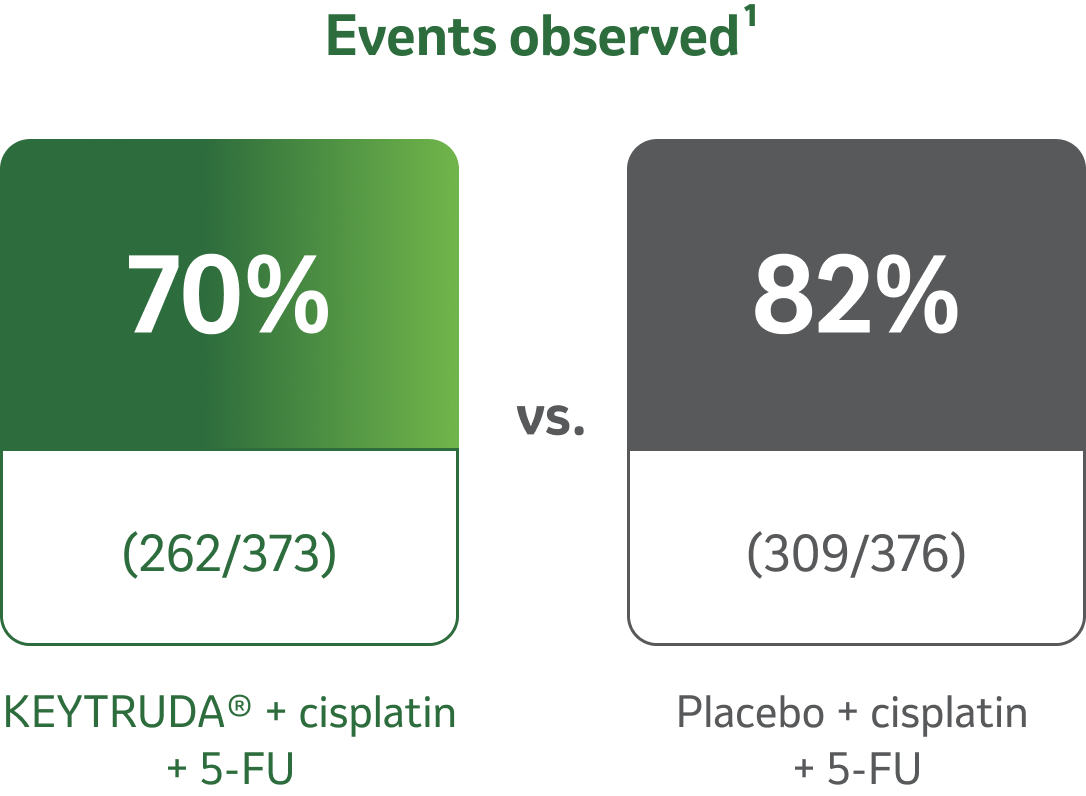
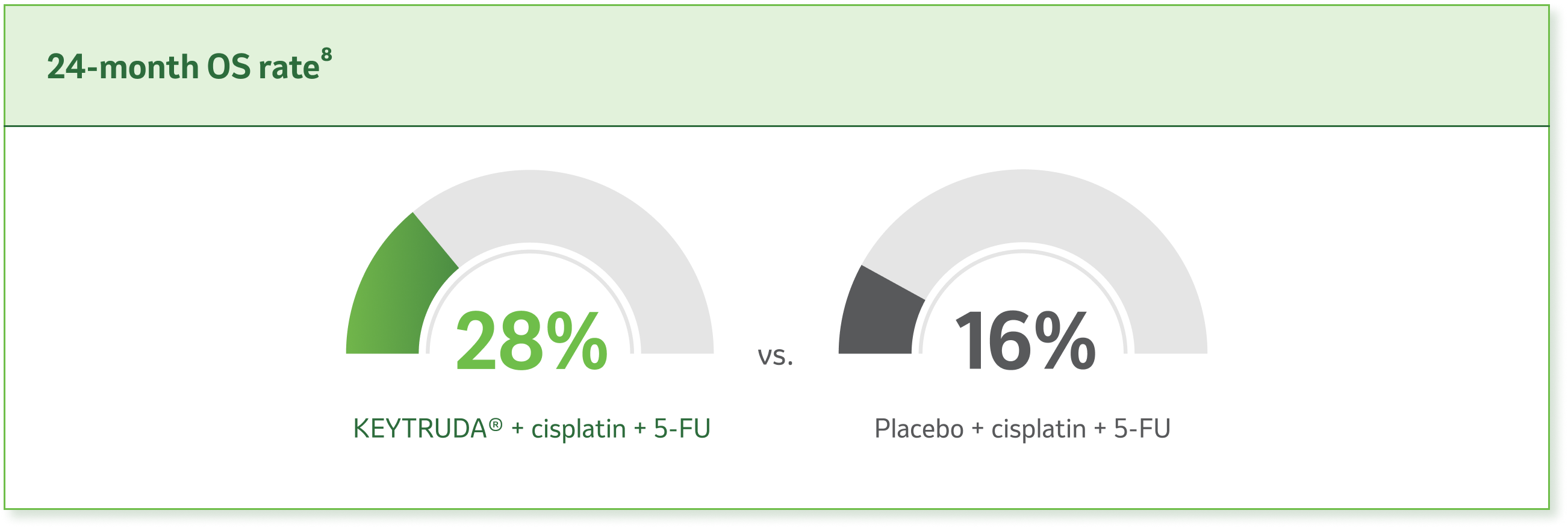
A substantial extension in OS was demonstrated KEYTRUDA® + cisplatin + 5-FU combination therapy vs. placebo + cisplatin + 5-FU8

Median follow-up time: 22.6 months (IQR 19.6–27.1)8

reduction in the risk of disease progression or death8
HRd=0.73 (95% CI, 0.62–0.86; pe<0.0001) in all randomized patients

dBased on the stratified Cox proportional hazard model.8
eBased on a stratified log-rank test.8
fBased on Kaplan-Meier estimation.8
5-FU: 5-fluorouracil; CI: confidence interval; HR: hazard ratio; IQR: interquartile range; PFS: progression-free survival

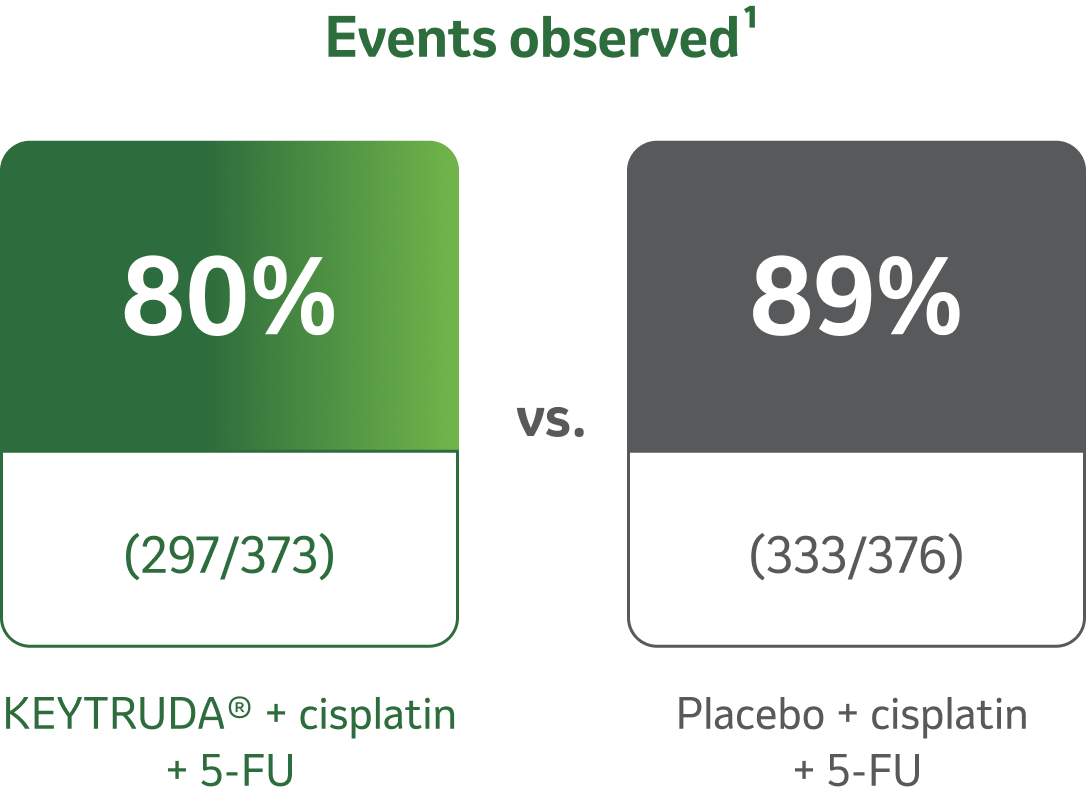
Significant improvement in PFS was observed with KEYTRUDA® + cisplatin + 5-FU vs. placebo + cisplatin + 5-FU8

Median follow-up time: 22.6 months (IQR 19.6–27.1)8

reduction in the risk of disease progression or death8
HRd=0.65 (95% CI, 0.55–0.76; pe<0.0001) in all randomized patients


dBased on the stratified Cox proportional hazard model.8
eBased on a stratified log-rank test.8
fBased on Kaplan-Meier estimation.8
5-FU: 5-fluorouracil; CI: confidence interval; HR: hazard ratio; IQR: interquartile range; PFS: progression-free survival

Median follow-up time: 22.6 months (IQR 19.6–27.1)8

HIGHER ORR
with KEYTRUDA® + cisplatin + 5-FU compared with cisplatin + 5-FU alone
pg<0.0001 (95% CI, 9.0–22.5)8,14
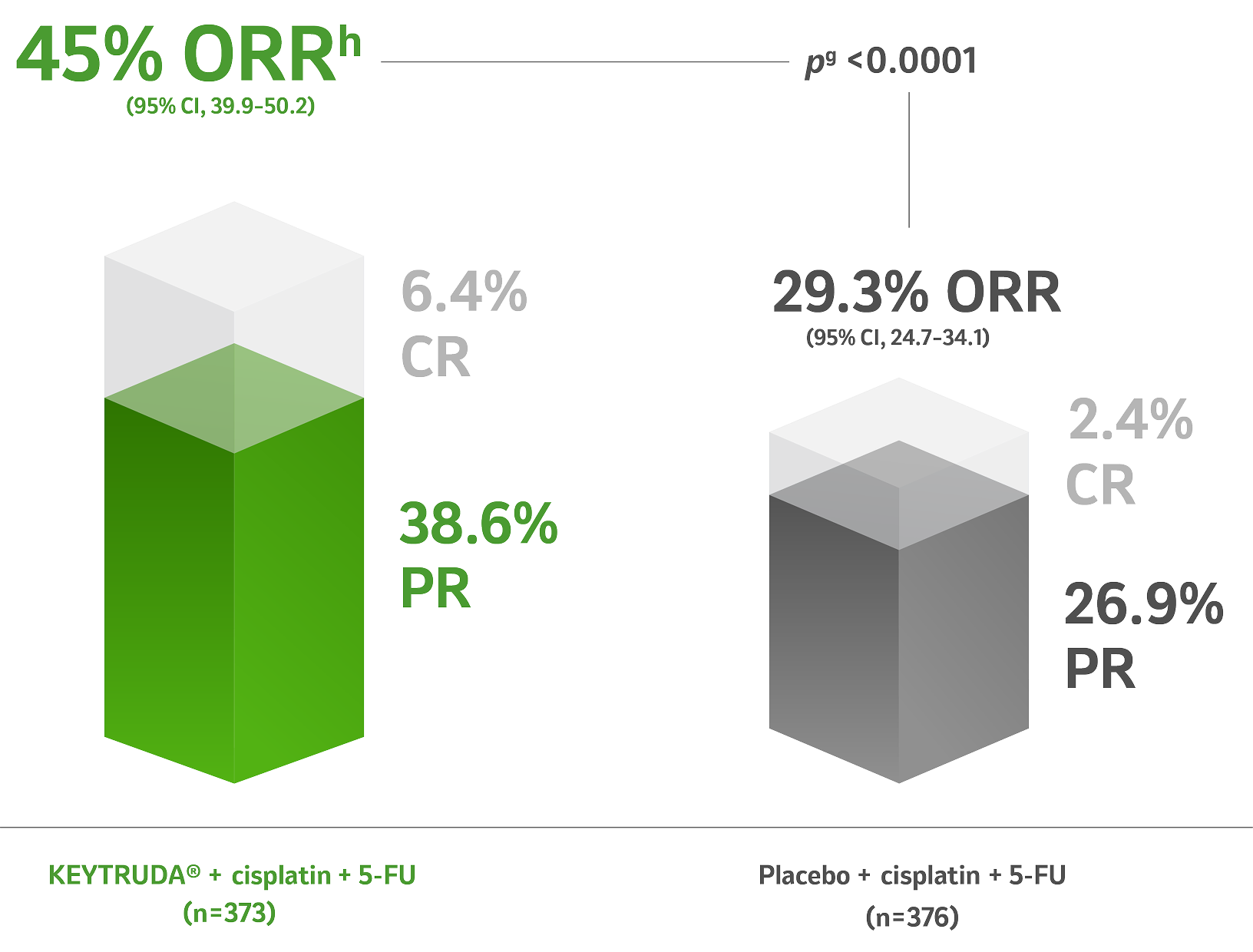

+ indicates that there was no progressive disease at the time of the last disease assessment.8
gOne-sided p value for testing.14
hBased on patients with a best overall response as confirmed complete or partial response.14
iBased on Kaplan Meier method for censored data.14
5-FU: 5-fluorouracil; CI: confidence interval; CR: complete response; DOR: duration of response; IQR: interquartile range; ORR: objective response rate; PR: partial response.
Study design: The efficacy of KEYTRUDA® was investigated in KEYNOTE-590, a multicenter, randomized, placebo-controlled trial that enrolled 749 patients. PD-L1 status was centrally determined in tumor specimens in all patients using the PD-L1 IHC 22C3 pharmDx kit.
Patients were randomized (1:1) to one of the following treatment arms; all study medications were administered via intravenous infusion:
-
KEYTRUDA® 200 mg on Day 1 of each 3-week cycle in combination with cisplatin 80 mg/m2 IV on Day 1 of each 3-
week cycle for up to six cycles and FU 800 mg/m2 IV per day on Day 1 to Day 5 of each 3-week cycle, or per local
standard for FU administration, for up to 24 monthss -
Placebo on Day 1 of each 3-week cycle in combination with cisplatin 80 mg/m2 IV on Day 1 of each three-week cycle
for up to six cycles and FU 800 mg/m2 IV per day on Day 1 to Day 5 of each 3-week cycle, or per local standard for FU
administration, for up to 24 months.
Treatment with KEYTRUDA® or chemotherapy continued until unacceptable toxicity or disease progression. Patients could be treated with KEYTRUDA® for up to 24 months in the absence of disease progression.
Primary endpoint: OS and PFS as assessed by the investigator according to RECIST-v1.1 (modified to follow a
maximum of 10 target lesions and a maximum of 5 target lesions per organ).
Secondary endpoint: ORR and DOR, according to modified RECIST-v1.1, as assessed by the investigator.
Inclusion criteria: Patients with metastatic or locally advanced esophageal or gastroesophageal junction (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma who were not candidates for surgical resection or definitive chemoradiation.
Exclusion criteria: Patients with active autoimmune disease, a medical condition that required immunosuppression, or who received prior systemic therapy in the locally advanced or metastatic setting were ineligible.
Results: The trial demonstrated a statistically significant improvement in OS and PFS for patients randomized to KEYTRUDA® in combination with chemotherapy, compared to chemotherapy.
DOR: duration of response; FU: fluorouracil; GEJ: gastroesophageal junction; IHC: immunohistochemistry; IV: intravenous; ORR: objective response rate; OS: overall survival; PD-L1: programmed death ligand 1; PFS: progression-free survival; RECIST: Response Evaluation Criteria in Solid Tumor

Choose between two dosing options for KEYTRUDA®, and your choice of platinum-/fluoropyrimidine-based chemotherapy1
Possible chemotherapy backbones for locally advanced unresectable or metastatic esophageal tumors in combination with KEYTRUDA®1,12
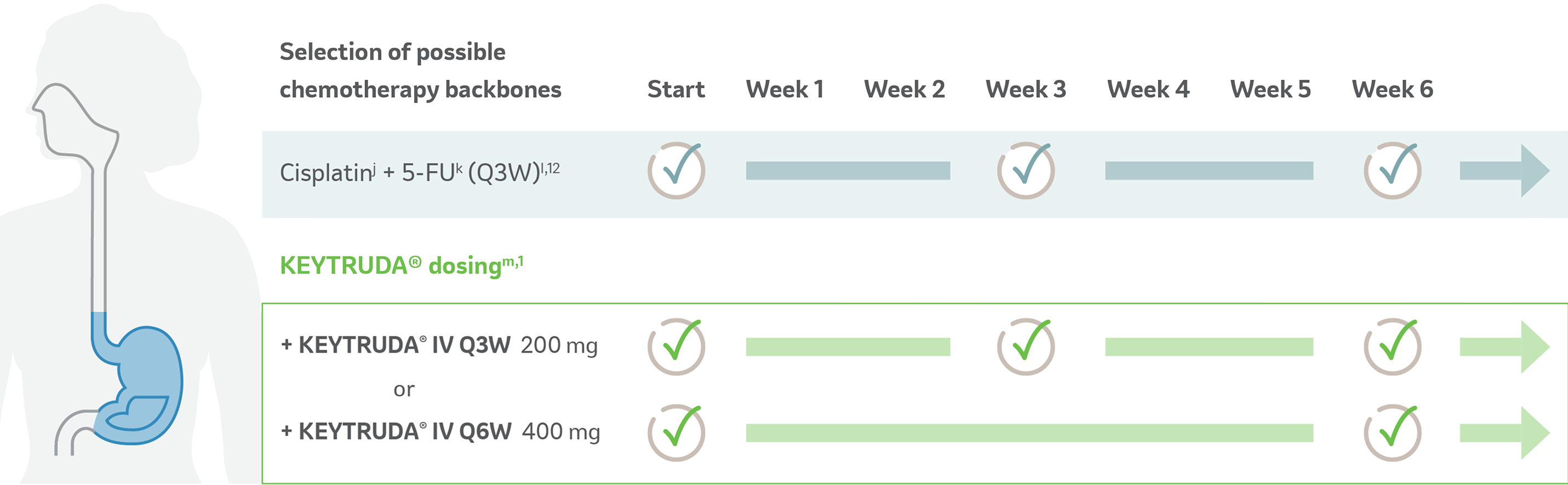
Please note:
- The KEYNOTE-590 trial investigated the combination of KEYTRUDA® + chemotherapy (cisplatin + 5-FU) Q3W for up to 35 cycles. All treatments were administrated until disease progression or unacceptable toxicity.1,8
- KEYTRUDA® 200 mg with cisplatin 80 mg/m2 IV on Day 1 of each 3-week cycle for up to 6 cycles + 5-FU 800 mg/m2 IV per day on Day 1 to Day 5 of each 3-week cycle (or per local standard for 5-FU administration) for up to 24 months.1
- Administer KEYTRUDA® prior to chemotherapy when given on the same day.1
jDay 1 of each 3 week cycle for up to six cycles.1
kDay 1 to Day 5 of each 3 week cycle, or per local standard for 5 FU administration.1
lCisplatin 80 mg/m2 IV on Day 1, fluorouracil 800 mg/m2 IV continuous infusion over 24 hours daily on Days 1–5; cycled every 21 days.12
mKEYTRUDA® treatment should be continued until disease progression or unacceptable toxicity, or up to 24 months.1
5-FU: 5-fluorouracil; IV: intravenous; Q3W: every 3 weeks; Q6W: every 6 weeks.
KEYTRUDA® (Pembrolizumab) Selected Safety Information (SSI)
Selected indications: Treatment of patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation, either in combination with platinum- and fluoropyrimidine based chemotherapy or as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by a validated test.
Dosage & Administration: KEYTRUDA® is administered 30 minutes intravenous infusion.
Combination Therapy: Adult patients with esophageal cancer is 200 mg every 3 weeks or 400 mg every 6 weeks (administer KEYTRUDA® prior to chemotherapy when given on the same day), until disease progression or unacceptable toxicity, or up to 24 months. Adult patients with HER2-positive is 200 mg every 3 weeks or 400 mg every 6 weeks (administer KEYTRUDA® prior to trastuzumab and chemotherapy when given on the same day), until disease progression, unacceptable toxicity, or up to 24 months.
Contraindications: None.
Precautions/Warnings: KEYTRUDA® can cause severe and fatal immune-mediated adverse reactions, can occur in any organ system or tissue, as immune-mediated pneumonitis, immune-mediated colitis, hepatotoxicity and immune-mediated hepatitis, immune-mediated nephritis with renal dysfunction, immune-mediated endocrinopathies (as adrenal insufficiency, hypophysitis, hyperthyroidism, hypothyroidism, thyroiditis & type 1 diabetes mellitus) & immune-mediated dermatologic adverse reactions. KEYTRUDA® can cause other immune-mediated adverse reactions as: myocarditis, pericarditis, vasculitis, meningitis, encephalitis, myelitis and demyelination, myasthenic syndrome/myasthenia gravis (including exacerbation), Guillain-Barré syndrome, nerve paresis, autoimmune neuropathy, pancreatitis, to include increases in serum amylase and lipase levels, gastritis, duodenitis, hypoparathyroidism, myositis/polymyositis, rhabdomyolysis (and associated sequelae, including renal failure), arthritis (1.5%), polymyalgia rheumatica, hemolytic anemia, aplastic anemia, hemophagocytic lymphohistiocytosis, systemic inflammatory response syndrome, histiocytic necrotizing lymphadenitis (Kikuchi lymphadenitis), sarcoidosis, immune thrombocytopenic purpura, solid organ transplant rejection, other transplant (including corneal graft) rejection, uveitis, iritis and other ocular inflammatory toxicities can occur. Some cases can be associated with retinal detachment. Various grades of visual impairment, including blindness, can occur. If uveitis occurs in combination with other immune-mediated adverse reactions, consider a Vogt-Koyanagi-Harada-like syndrome, as this may require treatment with systemic steroids to reduce the risk of permanent vision loss. KEYTRUDA® can cause severe or life-threatening infusion-related reactions, including hypersensitivity and anaphylaxis. KEYTRUDA® can cause complications of allogeneic HSCT. Increased mortality in patients with multiple myeloma when KEYTRUDA® is added to a thalidomide analogue and dexamethasone. KEYTRUDA® can cause fetal harm when administered to a pregnant woman.
Adverse Reactions: Most common adverse reactions (reported in ≥20% of patients) were:
- KEYTRUDA® as a single agent: fatigue, musculoskeletal pain, rash, diarrhea, pyrexia, cough, decreased appetite, pruritus, dyspnea, constipation, pain, abdominal pain, nausea, and hypothyroidism.
- KEYTRUDA® in combination with chemotherapy or chemoradiotherapy: fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, pyrexia, alopecia, peripheral neuropathy, mucosal inflammation, stomatitis, headache, weight loss, abdominal pain, arthralgia, myalgia, insomnia, palmar-plantar erythrodysesthesia, urinary tract infection, and hypothyroidism.
Please refer to the full prescribing information of KEYTRUDA® for more information.
CPS: combined positive score; FU: fluorouracil; HNSCC: head and neck squamous cell carcinoma; HST: hematopoietic stem cell transplantation; PD-L1: programmed death ligand 1.


Expert discussion: 6+ years of high-risk eTNBC survival data
In an exclusive interview, Prof. Schmid reveals the significant survival outcomes from the 7th interim analysis of the KEYNOTE-522 study.

Extend survival for your patients
KEYTRUDA® combination therapy: your first-line choice for appropriate patients. Explore KEYNOTE-590 data and its impact on clinical outcomes.

Redefine cancer care with an MDT
Put your patients first with an MDT. Discover how a multidisciplinary approach can improve your patients’ outcomes.
References:
1. KEYTRUDA®. Prescribing Information. 2. Rischin D, Harrington KJ, Greil R, et al. Pembrolizumab alone or with chemotherapy for recurrent or metastatic head and neck squamous cell carcinoma: Health-related quality-of-life results from KEYNOTE-048. Oral Oncol. 2022;128:105815. 3. Rhoten BA, Murphy B, Ridner SH. Body image in patients with head and neck cancer: a review of the literature. Oral Oncol. 2013;49(8):753-60. 4. So WK, Chan RJ, Chan DN, et al. Quality-of-life among head and neck cancer survivors at one year after treatment–a systematic review. Eur J Cancer. 2012;48(15):2391-408. 5. EuroQol–a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199-208. 6. Burtness B, Harrington KJ, Greil R, et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet. 2019;394(10212):1915-28. 7. Harrington KJ, Burtness B, Greil R, et al. Pembrolizumab with or without chemotherapy in recurrent or metastatic head and neck squamous cell carcinoma: Updated results of the phase III KEYNOTE-048 study. J Clin Oncol. 2023;41(4):790-802. 8. Burtness B, Harrington KJ, Greil R, et al. Supplementary appendix: Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. The Lancet. 2019;394(10212):1915-28.
AE-KEY-00951 I Expiry date: 30.06.2027