KN-052



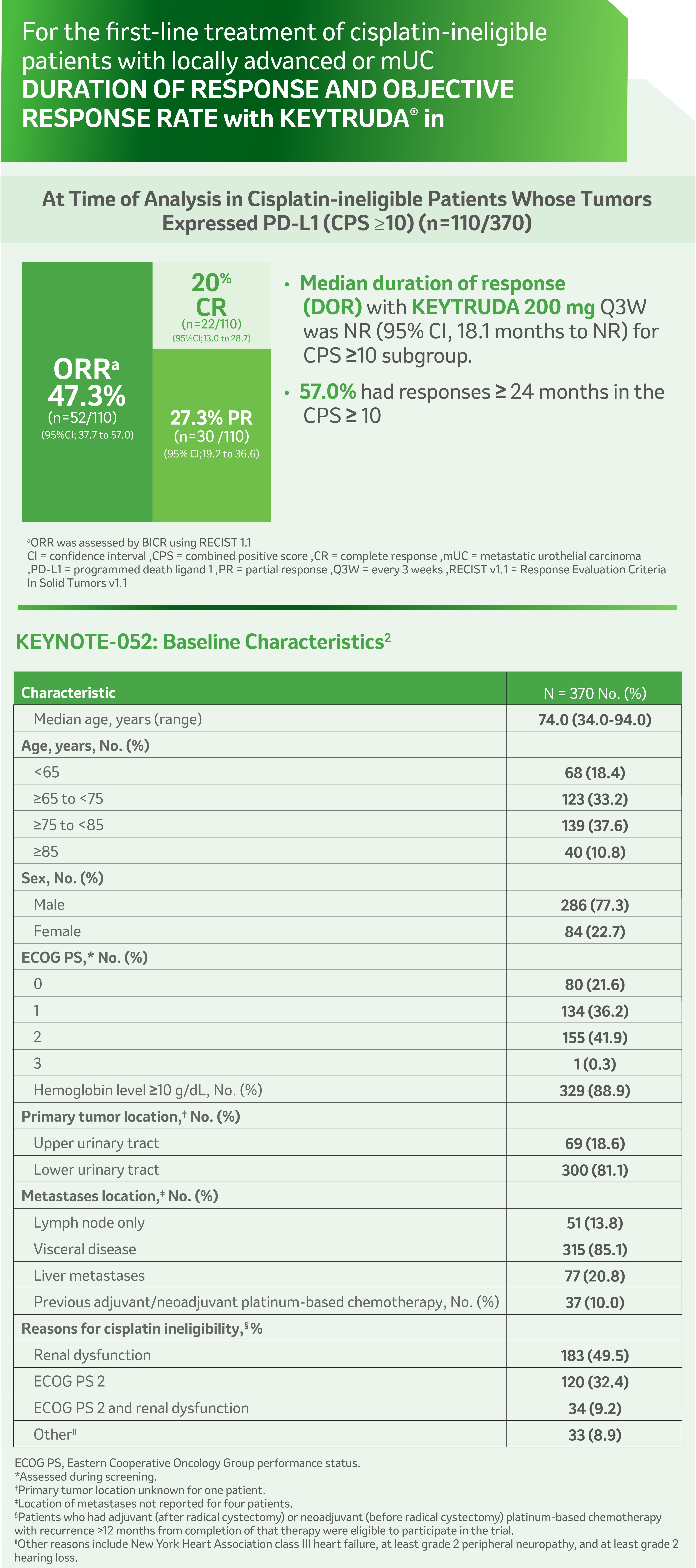

References:
- Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology,38(23), 2658–2666. https://doi.org/10.1200/JCO.19.01213
- Supplementary of Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23), 2658– 2666. https://doi.org/10.1200/JCO.19.01213
- Balar, A.V. et al. (2017) ‘First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (Keynote-052): A Multicentre, single-arm, phase 2 study’, The Lancet Oncology, 18(11), pp. 1483–1492. doi:10.1016/s1470-2045(17)30616-2








References:
- Balar, A. V., Castellano, D. E., Grivas, P., Vaughn, D. J., Powles,., & O’Donnell, P. H. (2023). Efficacy and safety of pembrolizumab in metastatic urothelial carcinoma: results from KEYNOTE-045 and KEYNOTE-052 after up to 5 years of follow-up. Annals of oncology : official journal of the European Society for Medical Oncology, 34(3), 289–299.https://doi. org/10.1016/j.annonc.2022.11.012
- Supplementary of Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23), 2658– 2666. https://doi.org/10.1200/JCO.19.01213
- Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23),2658–2666. https://doi.org/10.1200/JCO.19.01213
- Balar, A.V. et al. (2017) ‘First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (Keynote-052): A Multicentre, single-arm, phase 2 study’, The Lancet Oncology, 18(11), pp. 1483–1492. doi:10.1016/s1470-2045(17)30616-2
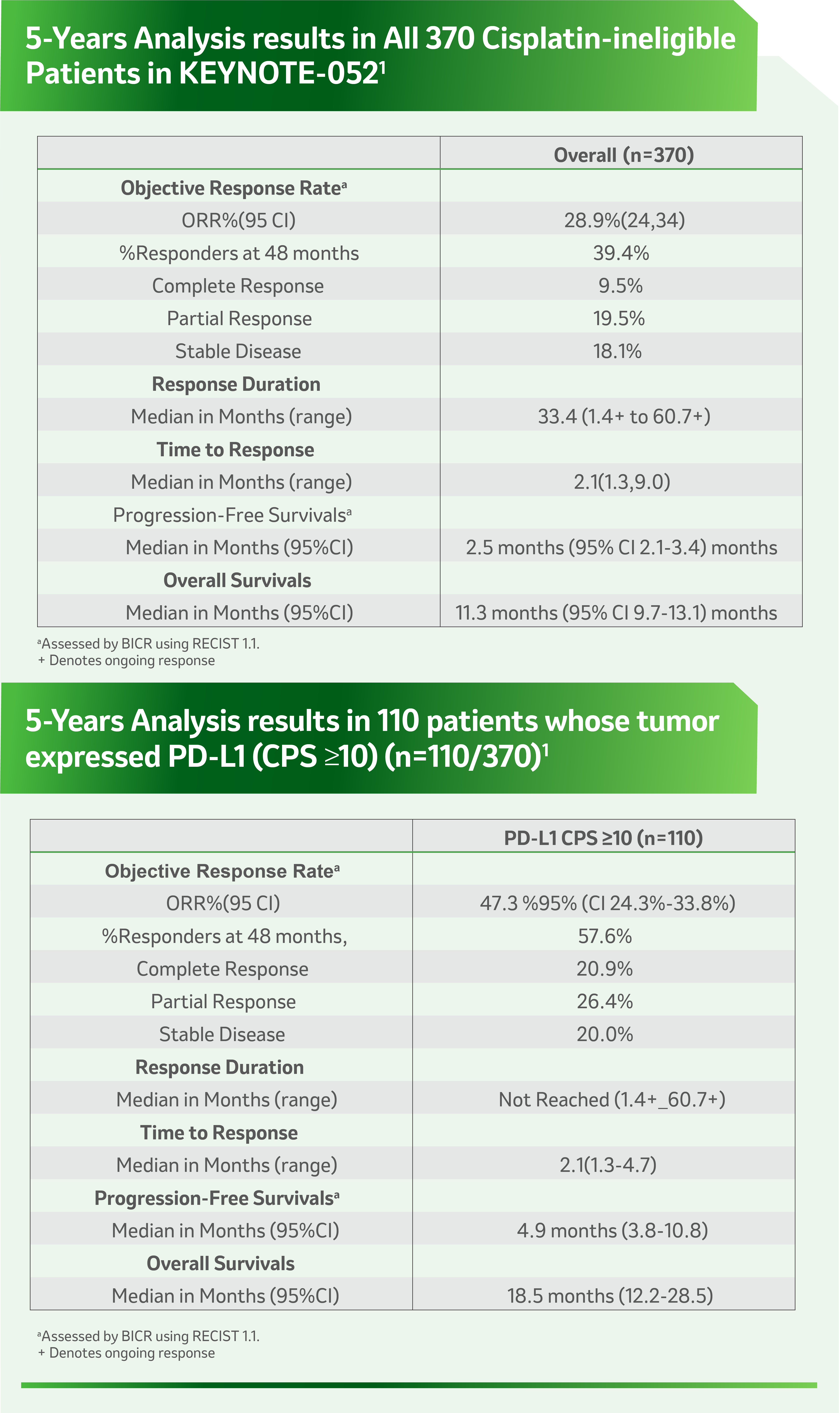
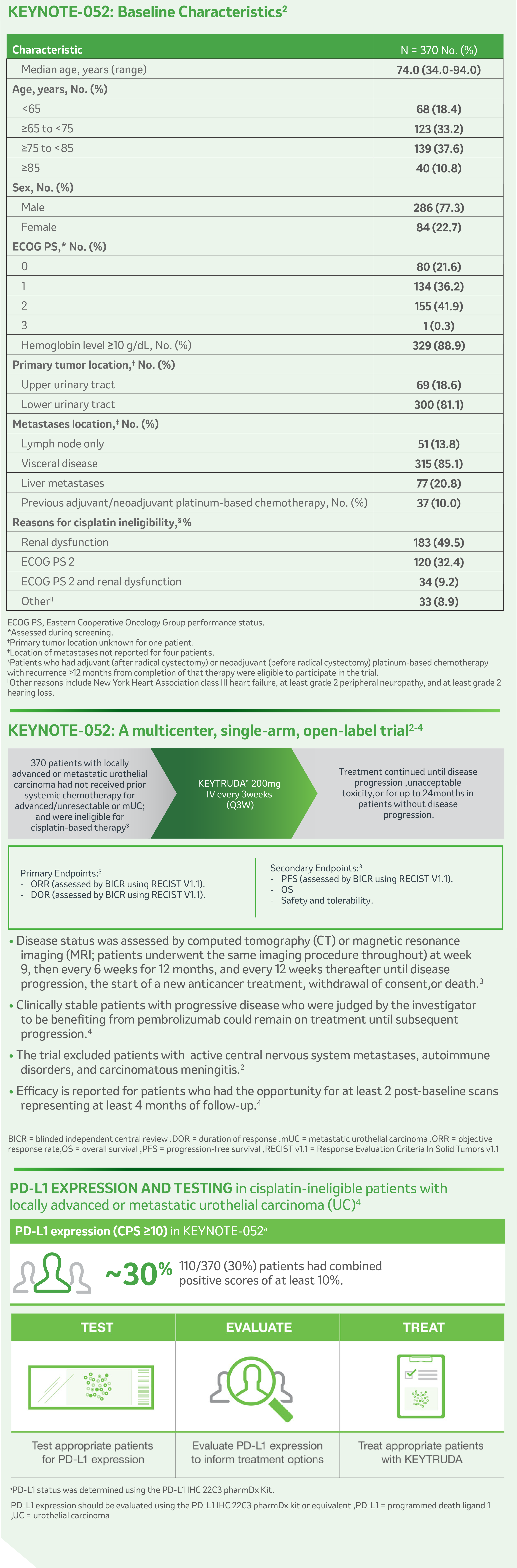
References:
- Balar, A. V., Castellano, D. E., Grivas, P., Vaughn, D. J., Powles,., & O’Donnell, P. H. (2023). Efficacy and safety of pembrolizumab in metastatic urothelial carcinoma: results from KEYNOTE-045 and KEYNOTE-052 after up to 5 years of follow-up. Annals of oncology : official journal of the European Society for Medical Oncology, 34(3), 289–299.https://doi. org/10.1016/j.annonc.2022.11.012
- Supplementary of Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23), 2658– 2666. https://doi.org/10.1200/JCO.19.01213
- Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23),2658–2666. https://doi.org/10.1200/JCO.19.01213
- Balar, A.V. et al. (2017) ‘First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (Keynote-052): A Multicentre, single-arm, phase 2 study’, The Lancet Oncology, 18(11), pp. 1483–1492. doi:10.1016/s1470-2045(17)30616-2
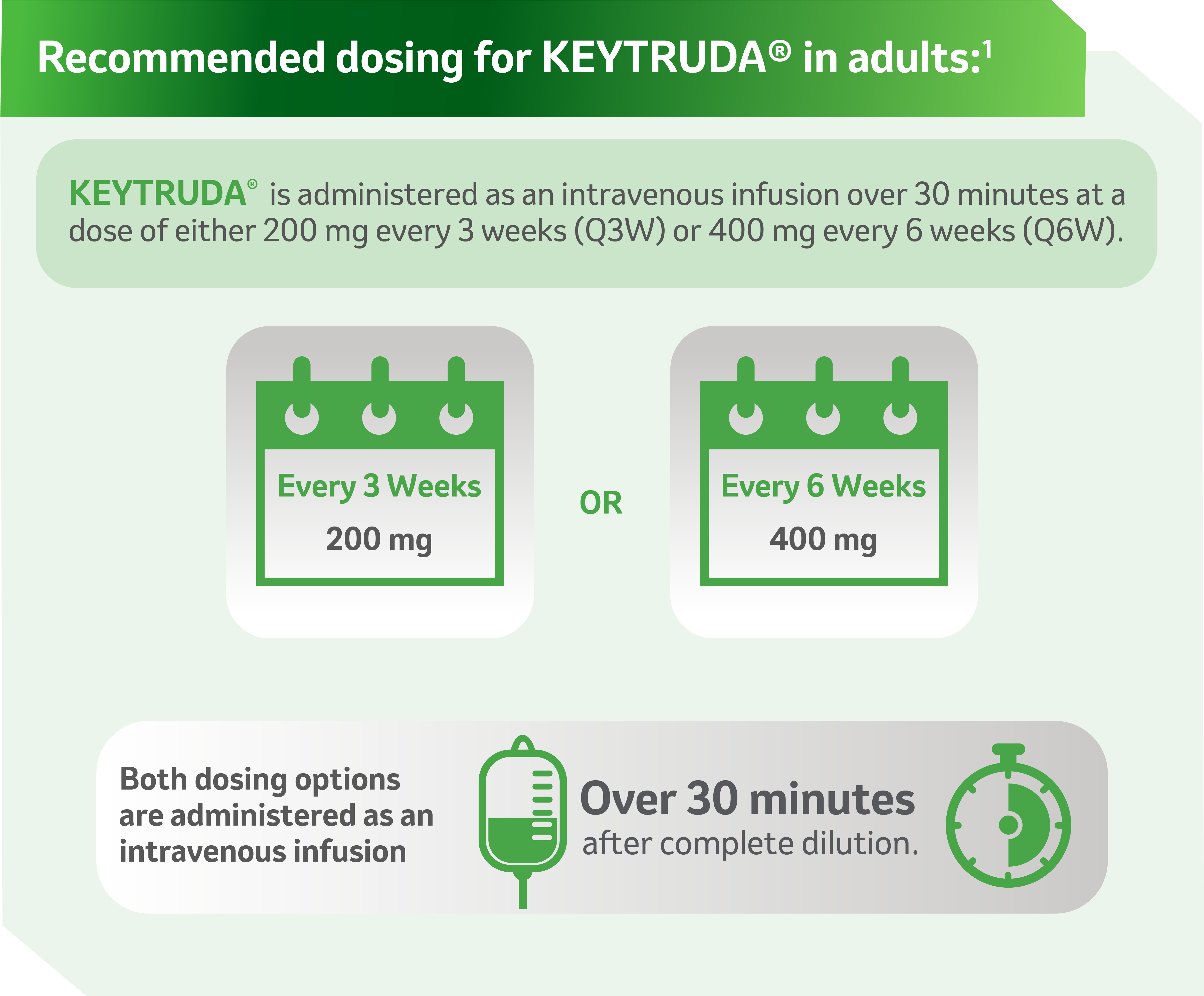
References:
- KEYTRUDA® Prescribing Information.


References:
- Balar, A. V., Castellano, D. E., Grivas, P., Vaughn, D. J., Powles,., & O’Donnell, P. H. (2023). Efficacy and safety of pembrolizumab in metastatic urothelial carcinoma: results from KEYNOTE-045 and KEYNOTE-052 after up to 5 years of follow-up. Annals of oncology : official journal of the European Society for Medical Oncology, 34(3), 289–299.https://doi. org/10.1016/j.annonc.2022.11.012
- Supplementary of Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23), 2658– 2666. https://doi.org/10.1200/JCO.19.01213
- Vuky, J., Balar, A. V., Castellano, D., O’Donnell, P. H., Grivas, (2020). Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 38(23),2658–2666. https://doi.org/10.1200/JCO.19.01213
- Balar, A.V. et al. (2017) ‘First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (Keynote-052): A Multicentre, single-arm, phase 2 study’, The Lancet Oncology, 18(11), pp. 1483–1492. doi:10.1016/s1470-2045(17)30616-2
KW-OMB-00004 | Exp 15 Jun 2027